Circuitry and Brain-Body Interactions | 2021
Gut-to-Brain Circuit Contributions to Parkinson-Like Phenotypes in Disease Models
Study Rationale: Although clinicians have long reported that Parkinson’s disease (PD) does not affect the brain alone, the field has only recently started to investigate gut dysfunction in experimental models of PD. Consequently, the anatomical and functional basis of gut-to-brain circuitry dysfunction in PD—such as network activity and dopamine signaling—remain poorly understood. Team Gradinaru will characterize circuit mechanisms underlying gut-to-brain disease spread and progression in the earliest appearance of Parkinson’s symptoms.
Hypothesis: Team Gradinaru’s hypothesis is that environmental and genetic factors impact the connections between neurons in the enteric nervous system (ENS), which regulates the gastrointestinal tract. This disruption may increase susceptibility to PD triggers—including aggregation of alpha-synuclein, which is toxic to cells and can trigger PD symptoms and gut inflammation. This inflammation could, in turn, augment the toxic form of alpha-synuclein that circulates to the brain and causes dysfunctions in neural circuits and motor deficits.
Study Design: Team Gradinaru will determine PD-relevant gut and brain anatomic and physiologic profiles in rodents, primates, and human cells by clarifying the anatomic pathways underlying gut-to-brain propagation of aggregated alpha-synuclein in mice, and by constructing anatomic and functional maps of macaque ENS and central nervous system PD-relevant circuits at single-cell resolution. The team also will test whether disruption of ENS circuitry mitigates gut-brain disease outcome and evaluate whether spiny mice (a rodent model that can repair damaged tissue) show protection from PD-related gut-brain degeneration.
Impact on Diagnosis: Using powerful new technologies across multiple PD-relevant model systems, this project could uncover novel circuit mechanisms that mediate symptoms, and embolden new therapeutic options to slow, halt, or perhaps reverse peripheral symptoms of PD.
Leadership
Project Outcomes
This project will use powerful sensor, actuator, and gene delivery technologies across multiple model systems to characterize the anatomical and functional bases of gut-brain circuitry dysfunction in PD, enabling new therapeutic options to slow, halt, or perhaps even reverse peripheral symptoms of PD. View Team Outcomes.
Team Outputs
Click the following icons to learn more about the team’s outputs:
Overall Contributions
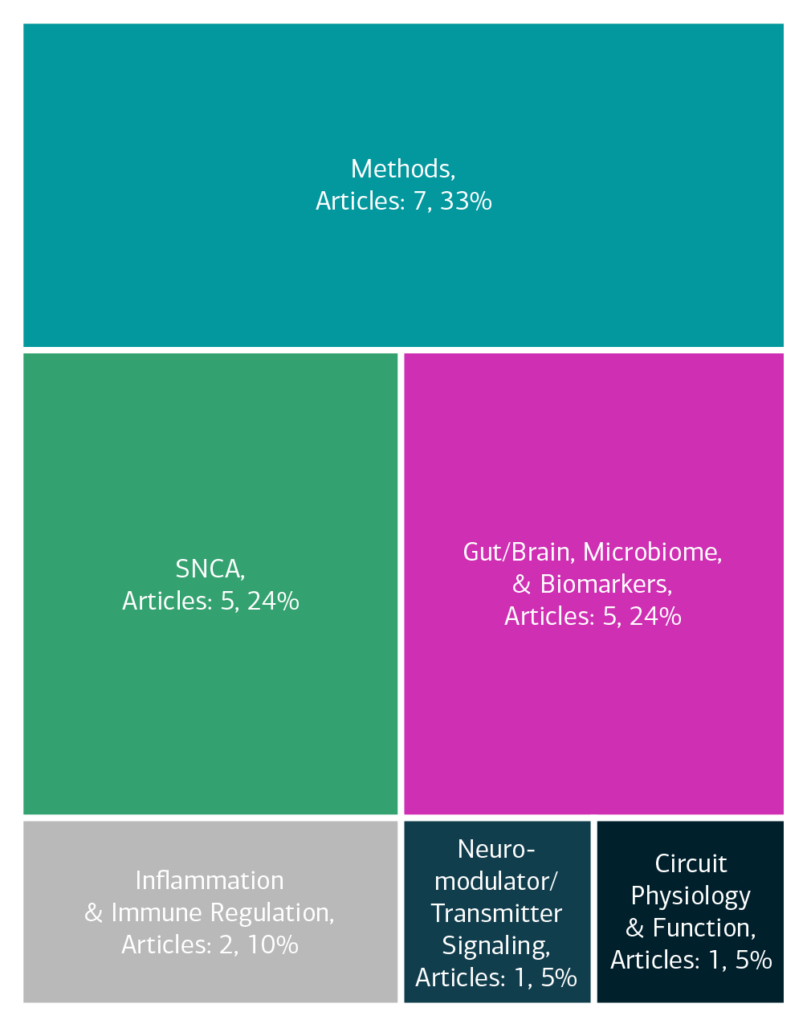
Here is an overview of how this team’s article findings have contributed to the PD field as of May 2025. There are two different categorizations of these contributions – one by impact to the PD community and a second by scientific category.
Impact

Category
Featured Output
Below is an example of a research output from the team that contributes to the ASAP mission of accelerating discoveries for PD.
Peripheral neuronal activation shapes the microbiome and alters gut physiology
Using viral vectors developed to specifically target the peripheral nervous system, transgenic mouse lines, and chemogenetic tools, the team assayed the effect of activating specific neuronal subpopulations associated with the gut. The resulting metagenomic, metabolomic, proteomic, and transcriptomic datasets provide a resource for interpreting the effects of ENS activation in mice. The team found differential effects of activating ChAT+ and TH+ neurons on gut physiology, with implications for PD, where damage to TH+ neurons is associated with constipation. In addition, the team assesses the broad effects of gut-associated neuronal activation on microbiome composition.
Team Accolades
Members of the team have been recognized for their contributions.
- Open Science Champions: Catherine Oikonomou, Gerard Coughlin
- Network Spotlights: Sarkis Mazmanian, Catherine Oikonomou
- Awards
- COSA Prize Winners 2022: Miguel Chuapoco (First Place)
- 2023 Collaborative Meeting: Jin Hyung Chung (First Place Poster Winner; San Diego)
Other Team Activities
- Protocol Particulars Interview: Production and Purification of AAVs with Damien Wolfe, MS
- Working Groups: Microbiome – Sarkis Mazmanian (Co-Chair)
- Interest Groups: Anatomical Tracing of Circuit Connectivity – Viviana Gradinaru (Chair)
In the News
- Caltech researchers team up to fight Parkinson’s disease (Caltech, October 26, 2021)
- A common marker of neurological diseases may play role in healthy brains (ScienceDaily, January 9, 2024)












