Grounded in Discovery: Accelerating Parkinson’s Research Across the Globe
Parkinson’s disease is the world’s second most common neurodegenerative disease and the fastest-growing in prevalence. Though existing therapies can effectively manage some symptoms in the earlier stages of the disease, there currently is no cure and no disease-modifying treatments that can prevent or slow the underlying neurodegeneration.
Aligning Science Across Parkinson’s (ASAP) is a global research initiative centered on accelerating scientific advances in Parkinson’s disease research. Unlike traditional funding programs, our model is built on making large, long-term commitments, embracing the elements of uncertainty necessary to support high-risk, high-reward studies, and fostering a collaborative and open approach to challenge the status quo.

Much of our work to date has focused on identifying research gaps and building the essential infrastructure to accelerate discoveries. We have diligently identified critical needs and established transparent, collaborative research processes and environments that incorporate input across sectors and disciplines.
This foundational work includes the development and expansion of our supported programs
An international, multidisciplinary, and multi-institutional network of collaborating investigators working to address high-priority research questions
An ambitious resource program to understand the genetic architecture of Parkinson’s disease through genotyping over 250,000 volunteers around the world
As the end of our fifth full year approaches, we are proud to share what we have built. The infrastructure and processes we have established are changing the way science is typically done and are now generating significant momentum. This report details how we are continuing to push the Parkinson’s disease research field forward, translating our strong foundation into a future of accelerated discoveries.
In 2025, we focused on four major objectives across the ASAP portfolio
Objective 1
Funnel new ideas into the Parkinson’s disease research and development pipeline. To do this, we focused efforts on enhancing our mechanistic and global understanding of Parkinson’s disease by building upon past discoveries and opening up new opportunities to join the initiative.
Objective 2
Facilitate the rapid exchange of scientific ideas. ASAP is focused on removing silos and ensuring that the discoveries and thought leadership from our initiative are actively shared. In 2025, we participated in convenings (both as host and guest) to spark new ideas and foster collaboration.
Objective 3
Ensure others can leverage ASAP-funded work. Our focus in 2025 centered on integrating ASAP datasets with existing tools to promote analyses, along with generating, characterizing, and validating new tools, including iPSCs, antibodies, and rodent models.
Objective 4
Establish a strong, diverse, and global pipeline of the next generation of Parkinson’s disease researchers. Each of our programs supports early career researchers, whether through training courses, workshops, or dependent care support.
Our work has been made possible by the partners, collaborators, researchers, and study participants who continue to work alongside us to advance our collective understanding of Parkinson’s disease. We are grateful to our management organization, Coalition for Aligning Science, and our implementation partner, The Michael J. Fox Foundation for Parkinson’s Research (MJFF). We would also like to thank the Sergey Brin Family Foundation for its generous support in funding this initiative.
Looking ahead to 2026, we are expanding our funded research programs and the tools and resources available to the Parkinson's disease community, all of which will drive the research field further. As part of this effort, we will announce two new CRN team cohorts (scientific and technical tracks) focused on dissecting mechanisms that contribute to Parkinson's disease heterogeneity and developing novel tools to support Parkinson's disease research. For GP2, we hit over 100,000 participants with our 11th data release, and anticipate the identification of numerous risk variants in the coming year. GP2 is building upon this momentum to expand its focus from genetics to functional genomics of Parkinson’s disease subtypes in a global context. We are also excited to continue supporting PPMI, which is enrolling individuals at earlier stages of the disease to validate novel biomarkers related to diagnostics and disease progression. These initiatives represent a concerted effort to broaden our research footprint.
We invite you to keep reading to explore the impact of our four objectives this year and our upcoming plans.
Sonya B. Dumanis, PhD
Randy Schekman, PhD
Cornelis Blauwendraat, PhD
Ekemini A. U. Riley, PhD
Program Specific Overview & Milestones
In the following sections, we outline some of the 2025 outputs from ASAP’s supported programs and highlight how these outputs ladder up to our objectives.
Objective 1: Funnel new ideas into the Parkinson’s disease research and development pipeline.
Objective 2: Facilitate the rapid exchange of scientific ideas.
The core of our mission is to fuel innovation across Parkinson’s research and development. By supporting programs that fill critical resource and knowledge gaps, we continue to accelerate discoveries that deepen our understanding of disease biology and move promising ideas closer to impact. Building on significant findings from the past five years, our 2025 initiatives are expanding those advances and laying the groundwork for the next generation of breakthroughs.
Highlighted below are examples of areas our CRN teams are exploring, from circuit level dysfunction to cellular vulnerability.
The Role of Neural Timing in Motor Symptoms of Parkinson’s Disease

Movement-related activity in the internal globus pallidus of the parkinsonian macaque (Kase et al., 2024).
Progressive loss of dopamine neurons in the substantia nigra pars compacta (SNc) is associated with motor symptoms of Parkinson’s disease. It is increasingly apparent that Parkinson’s pathology extends beyond the SNc; however, how these broader changes contribute to the motor deficits observed in Parkinson’s disease remains unclear. CRN Team Strick has uncovered changes to the timing and synchrony of activity in the globus pallidus internus (GPi) and primary motor cortex (M1) during movement in preclinical Parkinson’s models. These changes correlated with the severity of motor deficits, suggesting altered timing and synchrony in regions outside of the SNc may contribute to the motor symptoms of Parkinson’s. The team’s findings laying the groundwork for the development of Parkinson’s disease treatments that target these regions.
Contributions of Neuromelanin in Parkinson’s Disease

Outcomes of the PIGMO model (Chocarro et al., 2025).
Neuromelanin is a by-product of dopamine metabolism that progressively accumulates with age, primarily in the substantia nigra pars compacta (SNc), which degenerate preferentially in Parkinson’s. However, because neuromelanin is not present in rodents, its role in Parkinson’s disease pathogenesis has been understudied in preclinical animal models. Using a novel rodent model, known as PIGMO, that expresses neuromelanin, CRN Team Vila has revealed sexually dimorphic adaptations of nigral dopamine neuron excitability and development of non-motor symptoms following neuromelanin accumulation, which may underlie male-specific susceptibility to Parkinson’s. Importantly, Team Vila has also demonstrated the utility of neuromelanin-sensitive MRI to detect intracellular neuromelanin accumulation in both rodents and humans, suggesting this technique may be useful as a biomarker of Parkinson’s disease.
Novel Roles of LRRK2 in Pre- and Post-Synaptic Function

Gene Ontology analysis of proteins with at least one differentially regulated phosphopeptide in striatum from vehicle vs. MLi2-treated Anxa1iCre LRRK2G2019S mice (Chen et al., 2025).
Increased LRRK2 kinase activity is observed in both genetic and idiopathic Parkinson’s disease cases, suggesting aberrant LRRK2 activity is a common underlying cause of the disease. However, the role of LRRK2 in dopamine neuron function and the mechanism by which LRRK2 leads to cellular dysfunction in Parkinson’s remains unclear. CRN Team Awatramani has identified novel roles of LRRK2 activity in both pre- and post-synaptic function of dopamine neurons, with pathogenic LRRK2 hyperactivity disrupting synaptic pathways, pre-synaptic active zone organization in the most vulnerable dopamine neurons, and post-synaptic D2-receptor signaling in the striatum. These findings establish a novel role of LRRK2 in circuit dysfunction in Parkinson’s disease.
Negative Regulation of LRRK2-Rab Signaling Has Therapeutic Potential
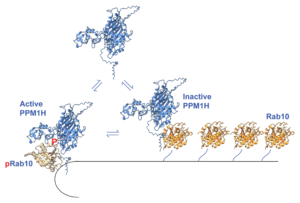
A model for PPM1H membrane recruitment and allosteric regulation (Adhikari et al., 2025).
LRRK2 plays a central role in lysosomal homeostasis by phosphorylating Rab GTPases. Pathogenic LRRK2 mutations enhance its kinase activity to phosphorylated Rab GTPases and lysosomal abnormalities, which are thought to contribute to the pathogenesis of Parkinson’s disease. CRN Team Alessi has identified two phosphatases – PPM1H and PPM1M – that are key, negative regulators of LRRK2-mediated Rab GTPase phosphorylation. These represent novel therapeutic targets for counteracting this LRRK2-driven hyperphosphorylation in Parkinson’s disease.
Bioenergetic Vulnerability of Dopamine Neurons
Dopamine neurons have large axonal arbors and vast numbers of synaptic terminals, imposing a substantial bioenergetic demand that may make them particularly vulnerable to stress and cell death. CRN Team De Camilli has identified that glycogen and triglycerides can meet the bioenergetic needs of synapses during neuronal activity. Notably, they found that glycogen availability is under neuromodulatory control by D2 receptors, such that dopamine itself provides a signal to store glycogen. If glycogen stores are depleted or access to them is blocked (like when dopamine neurons lose the ability to release dopamine), these cells become more sensitive to fuel restriction. These findings strongly support the idea that Parkinson’s disease has a bioenergetic component that could explain the preferential vulnerability of dopamine neurons.

Local glycogen storage levels in dopamine neurons are modulated by D2 auto-receptors (Pulido et al., 2025).

ASAP generated a significant number of findings this year. For a comprehensive look at what our investigators are learning, we invite you to explore the 2025 ASAP Research Roundup Newsletters from Q1, Q2, and Q3. Additionally, in 2025, we released a formal evaluation of the CRN’s impact in galvanizing the global research community. The resulting CRN Impact Report highlights findings from our 35 multidisciplinary, international, and collaborative teams across 14 countries.
GP2 is focused on deepening our understanding of Parkinson’s disease genetics within a global context. Below, we provide specific examples of GP2 findings around the world, including Africa, Asia, Europe, Latin America, and North America.
Capturing Information Around the World

The GP2 program has created a pipeline for conducting collective, global research at an unprecedented scale. In a recent podcast interview, ASAP Managing Director, Sonya Dumanis, and GP2 co-lead, Andrew Singleton, discussed the GP2 model. Listen in to learn more.
GP2 performed the largest ever multi-ancestry investigation to date, analyzing genetic data from nearly 70,000 individuals, with about 30% of non-European ancestry. Results indicate that ~2% of individuals with Parkinson’s disease carry a known high-risk variant, but this rate can vary dramatically between populations. For example, the prevalence of known causative variants is high in Ashkenazi Jewish (>12%) and Middle Eastern (>20%) ancestries but extremely low (<0.5%) in African, African-Admixed, and Central Asian populations.
Africa
Results from the first Parkinson’s disease genome-wide association study (GWAS) of South African and local African ancestry participants, posted earlier this year, revealed novel genetic insights into Parkinson’s pathogenesis.
Asia
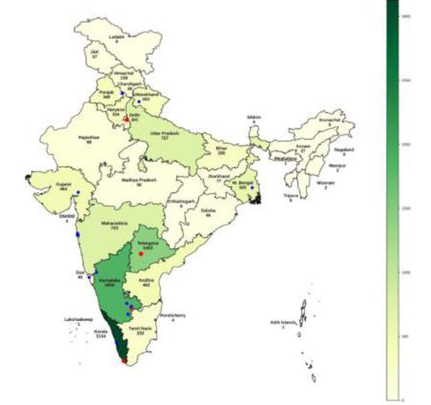
Pan-India recruitment of Parkinson’s disease cases and age, gender-matched controls (Kishore et al., 2025).
A pan-Indian genomic analysis of Parkinson’s disease revealed a striking overlap compared to Europeans while also identifying ancestry-specific genetic associations. These findings indicate an important role for genetic loci associated with immune function, lipid metabolism, and SNCA aggregation in Parkinson’s disease pathogenesis. As part of this study, India’s Parkinson’s biobank was established, creating the largest biobank in the South Asian region.
Assessment of cases from East Asian ancestry confirmed that the LRRK2 p.A419V is a significant Parkinson’s disease risk factor for these populations; and this variant is very rare in other populations of European, African American, African, Admixed American/Latin American, and Middle Eastern descent.
Europe
Meta-analyses of Parkinson’s disease GWAS study summary statistics showed new insights in Parkinson’s, with over 130 new risk loci identified in European populations. These findings expanded the number of known independent Parkinson’s disease-associated variants from 90 to 157, which is approximately a 74% increase.
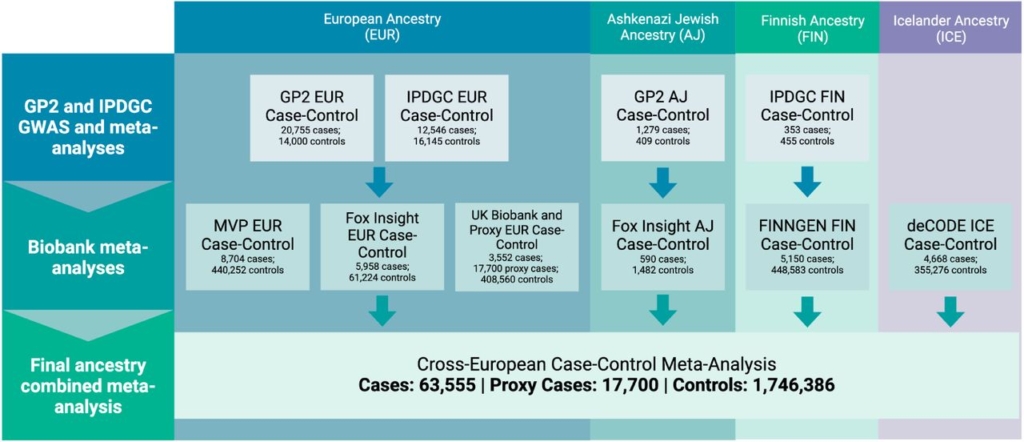
Flowchart detailing study design schema and sample counts (GP2 et al., 2025).
Latin America
The Latin American Research Consortium on the Genetics of Parkinson’s Disease (LARGE-PD) is a multicenter collaboration across Latin America in an effort to increase the knowledge about Parkinson’s in these countries. Analysis of 15 countries across the Americas and the Caribbean, the largest Latin-American GWAS study for Parkinson’s disease susceptibility, revealed Parkinson’s-associated variants in SNCA and ITPKB.

Manhattan plots from random-effects genome-wide association meta-analysis (GWAMA) of LARGE-PD Phase 1 and 2 cohorts (Leal et al., 2025).
North America
The GP2 cohort, Black and African American Connections to Parkinson’s Disease (BLAAC PD), which enrolled its 1,000th participant in August 2025, expanded to include both cognitive and smell testing.
PPMI serves as a biomarker discovery engine, pioneering studies to detect and track early disease. Multiple substudies are underway to ensure the work can be leveraged across multiple detection modalities — from digital tools to advanced imaging. Below are some examples of studies underway this year.
Refining DaTscan Analysis for Clinical Trials
Since 1995, dopamine transporter (DaT) imaging has been used in Parkinson’s disease research; however, high within-subject variability has limited its ability to measure longitudinal changes in clinical trials.
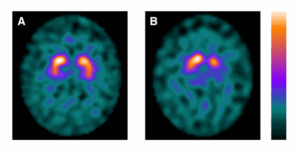
DAT SPECT imaging. Normal (a) and abnormal (b) [ 123 I] FP-CIT SPECT imaging of patients in the LEAP cohort. Patient A is a 64-year-old male. Patient B is a 63-year-old female. DAT, dopamine transporter; SPECT; single-photon emission computed tomography; LEAP, Levodopa in EArly Parkinson’s disease (Suwijn et al., 2019).
To address this challenge, PPMI re-analyzed more than 7,700 DaTscans across all timepoints using a new MIAKAT pipeline, which employs disease-specific templates, uses cerebral white matter as a reference region, and focuses on caudate and putamen subregions most susceptible to early disease changes. As showcased at the Parkinson’s Disease Therapeutics Conference in October, this improved analytical approach significantly enhanced the signal-to-noise ratio, improving statistical power to detect disease progression. This advancement promises to accelerate Parkinson’s therapeutic trials by enabling more sensitive detection of treatment effects with smaller sample sizes or shorter study durations.
Biochemical Biomarker Discovery Studies

PPMI Biomarkers Dashboard.
Examining biomarkers of neurodegenerative disease processes, including beta-amyloid, phosphorylated tau, NfL, and GFAP, have been conducted in previously collected plasma and CSF across all cohorts at early and later disease timepoints and analysis is underway. Additionally, these efforts are expanding, with results from unbiased omics approaches expected in the coming year. To stay up to date on activities in this area, check out the PPMI Biomarkers Dashboard.
Mapping Symptoms
PPMI seeks to identify the earliest signs of Parkinson’s disease and dementia with Lewy bodies through online surveys and virtual interviews. Participants rate the significance of health changes experienced over five years, creating “symptom maps” that could help clinicians recognize and intervene during prodromal stages.

Graphic showing the location of GAIT substudy wearable and mobile devices.
Digital Health Technology in Gait
This effort is using wearable and mobile devices to detect subtle movement changes that signal prodromal Parkinson’s disease. With approximately 275 participants enrolled and recruitment concluding by year’s end, the study will assess whether digital tools like Opal and Axivity sensors can identify and track early disease progression in real-world settings.
Imaging Studies
PPMI continues to advance PET imaging to validate novel tracers targeting inflammation (TSPO) and tau, while testing the ultra-high-resolution NeuroEXPLORER scanner. By comparing conventional and next-generation technology, researchers aim to define novel imaging biomarker signatures that predict disease onset and progression.
Learn more about PPMI substudies.
Below are examples across our programs on how we are continually building upon our prior work to maximize scientific and clinical impact.
Multi-Omics Meta-Analysis

Graphic symbolizing the Multi-Omics Meta-Analysis workflow.
Since the initiation of the ASAP CRN, teams have generated multiple large datasets using a diverse set of assays and techniques. Many CRN teams have generated complementary ‘omics datasets, including bulk RNAseq, single-cell RNAseq, ATACseq, and more. There is an urgent need to harmonize these datasets and look across them for replication, common patterns, and novel mechanistic insights only made possible through this scale and depth of data. In 2025, the CRN funded 10 projects to support meta-analysis across different CRN Cloud datasets.
From Genetic Variant Identification to Longitudinal Cohort Study

Overview of the African ancestry PD GBA1 GWAS locus (Jerez et al., 2024).
In 2023, GP2 announced a major breakthrough finding — the identification of a new GBA1 variant specific to Parkinson’s disease patients from West African populations. Additional assessment revealed that the variant leads to reduced GBA1 protein levels and overall activity, which indicated that this variant could serve as a potential therapeutic target. To fully understand the impact of this variant, GP2 is now initiating a longitudinal study to understand the impact of this variant on disease progression.
Shifting the Timeline: Getting Ahead of the Disease
PPMI is a longitudinal cohort study that aims to identify biomarkers of Parkinson’s risk, onset, and progression. It was launched in 2010 by The Michael J. Fox Foundation for Parkinson’s Research (MJFF). In 2020, ASAP provided funds to support an expansion of the study to more than 4,000 volunteers, including 2,000 prodromal participants recruited at nearly 50 international sites. This expansion was instrumental to a 2023 landmark study within PPMI that validated a new Parkinson’s biomarker that can detect early signs of Parkinson’s prior to clinical symptoms. This year, PPMI lowered its screening age to those 40 or older, enabling us to better understand what’s happening at early stages of the disease and uncover new prevention strategies.
Translating Functional Biology into the Therapeutic Promise

Three CRN researchers — Miratul Muqit, PhD (Team Alessi), Timothy Ryan, PhD (Team De Camilli), and James Surmeier, PhD (Team Surmeier) — were awarded SPARK-NS grants in 2025 to support the translation of their respective CRN findings into novel therapeutics for Parkinson’s.
In 2025, we expanded our research infrastructure by supporting new research efforts.
Dissecting the Mechanisms of Parkinson’s Disease Heterogeneity
In the Parkinson’s disease community, there is a saying that, “if you’ve met one person with Parkinson’s, you’ve met one person with Parkinson’s.” This adage underscores the complexity of Parkinson’s disease and highlights that every individual’s experience with the disease is unique from presentation to progression to treatment. In 2025, the CRN launched the Scientific Track funding opportunity, focusing on dissecting the mechanisms that contribute to Parkinson’s disease heterogeneity across six key focus areas: aging, co-pathologies, environment, circuit biology, clearance mechanisms, and alpha-synuclein seeding. Application reviews are currently underway, and we look forward to announcing funded teams in 2026.

CRN Scientific Track heterogeneity focus areas.
Developing Novel Tools for Emerging Targets
A thorough investigation of CRN and GP2 discoveries from the past five years revealed emerging targets, which could prove to be potential therapeutic targets for the research and development pipeline. Yet, the tools needed to validate these targets are severely lacking in some instances. To address this need, ASAP launched the CRN Technical Track funding opportunity, which focuses on supporting the development of novel tools to enable researchers to validate emerging targets to funnel them into the research and development pipeline. Application reviews are currently underway, and we look forward to announcing funded teams in 2026.

CRN Technical Track objectives and approaches.
Unlocking Brain Tissue Secrets Through Digital Innovation

For decades, researchers have faced a fundamental challenge. While brain tissue examination provides ground truth, access to high-quality, well-characterized samples has been severely limited, slowing progress in understanding the biological complexity of neurodegenerative conditions. In 2025, ASAP partnered with MJFF and The 10,000 Brains Project to pilot Path-ND, a digital brain banking program that digitizes brain tissue slides from Parkinson’s disease and other neurodegenerative diseases. This cloud-based FAIR data repository will make at least 1,000 combined Parkinson’s and healthy control cases accessible to researchers worldwide, including cases from ASAP’s Pilot Multi-site Slide Digitization Project. By democratizing access to brain tissue data, Path-ND enables identification of new mechanisms and disease subtypes — critical for Parkinson’s, where clinical diagnosis has a 10-30% misdiagnosis rate and co-occurring pathologies are common. AI analysis of this unprecedented dataset could fundamentally reshape our understanding of neurodegeneration, revealing interconnections across diseases and creating opportunities to rethink diagnostic categories and uncover novel therapeutic targets.
GP2 Expansion
Parkinson’s disease as a whole comprises mechanistically distinct processes, the importance of which varies between individuals and between stages of disease. To further resolve these processes, GP2 is expanding its focus from genetics to functional genomics of Parkinson’s disease subtypes in a global context. This expansion will enable the identification and mapping of distinct molecular mechanisms underlying Parkinson’s disease, while placing those in a cellular and temporal context.
Progress in Parkinson’s research depends heavily on individual discoveries and on the connections made between them. This year, we continued to bring scientists, clinicians, and collaborators together, bridging disciplines, sharing insights, and identifying new opportunities to close knowledge gaps. Through meetings and shared platforms, our network created momentum for the pursuit of better understanding and better outcomes for people with Parkinson’s.
ASAP is a research initiative that relies on collaborations with researchers and participants worldwide to uncover the roots of Parkinson’s disease. Currently, ASAP-affiliated research extends across 100 countries. Check out the map below to view the current ASAP community landscape.

Map showing the locations of ASAP’s resource program locations: GP2 cohorts, CRN investigators, and PPMI clinical sites, both combined and for each program separately.
PPMI invested in robust infrastructure to ensure that its digital tools could support remote collection at international sites.

myPPMI portal
myPPMI, a web-based portal for participants in the Parkinson’s Progression Markers Initiative, has been deployed to European sites participating in PPMI. All members of myPPMI are also enrolling in the broader PPMI Online study.
PPMI Online, an observational study collecting participant-reported information from people with and without Parkinson’s disease, has expanded beyond the United States to include Canada and the United Kingdom.
In keeping with prior years, all of the ASAP supported programs hosted Investigator Meetings, which brought together researchers from their respective programs to showcase findings and discuss the current Parkinson’s disease landscape.
CRN
Nearly 400 researchers from all 35 CRN teams convened at the 2025 CRN Collaborative Meeting in Phoenix, Arizona, sparking a dynamic exchange of ideas and partnership-building. The event showcased groundbreaking research in Parkinson’s disease, from functional genomics to neuro-immune interactions to circuitry, while amplifying the voices of patients and advocates who inspire the scientific community. Trainees shared their findings, and 11 trainees from CRN teams were honored for their research contributions.

2025 CRN Collaborative Meeting group photo.
As of November 2025, 34 interest group meetings were held with over 95 presentations from teams across the network. Interest group meetings are important forums to exchange pilot data and unpublished work. 100% of interest group meetings in 2025 featured preliminary findings. Trainees presented their findings at 94% of interest group meetings.
GP2
In March 2025, Black and African American Connections to Parkinson’s Disease (BLAAC PD) held its second Annual Meeting, convening investigators and coordinators from the study’s eleven active sites across the United States.

BLAAC PD second Annual Meeting group photo.
GP2 supported an African investigators meeting in Ghana to discuss unmet needs and opportunities with 20 attendees. This meeting led to the formation of the GP2 African Investigators Network (GAIN) and identified areas where additional training would be beneficial.
In 2023, GP2 supported a project that would enhance our understanding of Parkinson’s disease in Kyrgyzstan, representing the country’s first human genetics study of Parkinson’s disease. This support triggered a movement to bring neurologists and researchers side-by-side in workshops, webinars, and practice sessions. Now, two years later, these scientists are supporting patients and local healthcare systems across the country, which has prompted a national initiative supported by the Kyrgyz government.

GP2’s first workshop in Bishkek, Kyrgyzstan, in March 2024.
GP2 held its Annual Investigators Meeting in Hawai’i in October 2025, connecting over 240 researchers from around the globe to discuss progress and future directions for GP2.

2025 GP2 Annual Investigators Meeting.
PPMI

2025 Annual Parkinson’s Progression Markers Initiative Investigators Meeting.
In April, more than 300 researchers, clinicians, and study participants came together in New York City for the 2025 Annual Parkinson’s Progression Markers Initiative Investigators Meeting.
The event highlighted major advances in Parkinson’s disease research, including new diagnostic tools, expanded global screening, and refined biological staging to accelerate precision medicine.
ASAP engages the broader research community through scientific meetings and educational programming designed to disseminate findings and catalyze new collaborations.
In 2025, CRN Team Alessi hosted 11 LRRK2 Central Webinar events to discuss recent work on the role of LRRK2 in Parkinson’s disease research. Similarly, CRN Team Hurley utilized their Mito911 webinar series to highlight the latest research in mitochondrial biology, hosting five sessions. Don’t miss out on future updates! Visit our events page to register for upcoming Mito911 and LRRK2 Central webinars. Visit our YouTube channel to watch select Mito911 sessions.

Team Hurley hosted Mito911 webinars (Left). Team Alessi hosted LRRK2 webinars (Right).
GP2 hosted a luncheon at the Asian and Oceanian Parkinson’s Disease and Movement Disorders Congress (AOPMC) in Tokyo, Japan, to share GP2’s overarching goals, highlight significant progress, and discuss new opportunities for researchers to engage and lead GP2 studies. Over 70 researchers attended, sparking valuable discussions about collaboration across the region and globally.

- Asian and Oceanian Parkinson’s Disease and Movement Disorders Congress (AOPMC) group photo.
GP2 presented their work at several conferences, including at Mission MSA, National Center for Excellence in Research on Parkinson’s Disease (NCER), and the International Congress of Parkinson’s Disease and Movement Disorders® (MDS). In addition, BLAAC-PD sites participated in health fairs and community events throughout the year to connect with community groups and facilitate enrollment.

- GP2 team members at the International Congress of Parkinson’s Disease and Movement Disorders® in Hawai’i.
PPMI gave more than 35 presentations this year at various research conferences, including at ADPD, American Academy of Neurology, Alzheimer’s Association International Conference, and MDS. A collection of all PPMI meeting abstracts can be found on the Zenodo community.

Robert Thibault, PhD, presenting at the 10th International Congress on Peer Review and Scientific Publication.
The ASAP Open Science Team described how we implement our open science policy at the 10th International Congress on Peer Review and Scientific Publication. Watch the video beginning at 3:36:30.
In addition, we sponsored in-person research conferences, including:
– International Symposium on Autophagy
– Development of the Dopaminergic System: From Stem Cells to Circuits
– Neurodegeneration: Linking Cellular Pathways to Therapeutic Strategies Keystone
– Grand Challenges in Parkinson’s Disease
Open Science White Paper
This fall, we released a white paper version of Investing in Open Science: Key Considerations for Funders. The paper details ASAP’s Open Science Policy and barriers, costs, and opportunities for funders within the biomedical research ecosystem.
Responding to the NIH RFI on Maximizing Research Funds by Limiting Allowable Publishing Costs
This fall, we responded to the National Institutes of Health’s (NIH) Request for Information (RFI) to address rising article processing charges (APCs) and closed access publications. Our solution calls for:
– Requiring preprints to permit no-cost access to research outputs
– Funding preprint infrastructure to ensure sustainability
– Supporting services that add real value (e.g., rigorous peer review, compliance checks) rather than blunt APC caps that may have unintended consequences
Ultimately, the NIH can catalyze a fundamental shift toward a fair, open, and efficient publishing ecosystem that maximizes taxpayer value by investing in the open science infrastructure already underway.
Our commitment to open science ensures that discoveries made through ASAP-funded research extend far beyond individual labs. By developing and sharing resources — from datasets and imaging tools to biosamples and standardized protocols — we empower the global research community to build upon this foundation. This year, we continued to expand access to these tools and encourage their use in new studies, including over 2,200 ASAP-funded research outputs, including datasets, code, lab materials, and protocols through our external Catalog. Doing so drives broader understanding and faster innovation in Parkinson’s disease.
We believe in ensuring that our datasets are accessible and actionable.
CRN

CRN Cloud and Allen Institute collaboration.
This year, we announced a new collaboration with the Allen Institute to help researchers unlock insights into Parkinson’s and other neurodegenerative diseases. Through this collaboration, we united Collaborative Research Network (CRN) Cloud data from 3 million human cells across 9 brain regions from individuals with Parkinson’s into the Allen Brain Cell (ABC) Atlas visualization tool, which already contains 6.4 million human cells — an increase of nearly 50%! Explore the expanded, open-access ABC Atlas.
 ASAP funded the transformation of GuPPy (Guided Photometry Analysis in Python) into a sustainable platform that will integrate fiber photometry data with Neurodata Without Borders (NWB) and the DANDI archive. This standardization will enable researchers across laboratories to directly compare and meta-analyze neural circuit activity in Parkinson’s disease models using open, reproducible data formats.
ASAP funded the transformation of GuPPy (Guided Photometry Analysis in Python) into a sustainable platform that will integrate fiber photometry data with Neurodata Without Borders (NWB) and the DANDI archive. This standardization will enable researchers across laboratories to directly compare and meta-analyze neural circuit activity in Parkinson’s disease models using open, reproducible data formats.
GP2

Map of GP2 cohorts around the globe.
The GP2 Data Release 11 marked the release of over 100,000 genotypes, one-third of which are from underrepresented populations, making it one of the largest and most globally representative Parkinson’s disease datasets to date. In addition, the release includes 38,000 whole genome sequencing data and more than 400,000 clinical data points. At the scale of 100,000 globally representative samples, researchers have the statistical power to uncover new genetic risk factors for Parkinson’s disease. Release 11, which can be accessed on AMP® PDRD, will enable further study into the genetics of Parkinson’s disease, including GP2-wide analyses that focus on significant questions across multiple cohorts.
GP2 partnered with Broad Institute to develop a GP2 Genome Browser. This genome browser allows users to display variant and gene-level data broken down by ancestry.
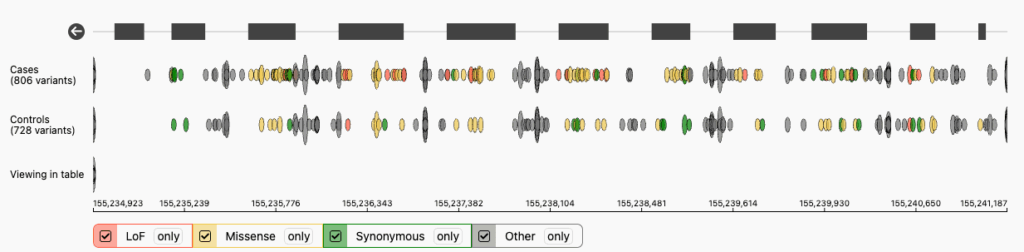
Example of GBA1 variant and gene-level data from individuals of diverse ancestries.
GP2 launched a collaboration between GP2, Parkinson’s Foundation, and Rune Labs to integrate genetic testing with wearable-based symptom tracking devices to identify genetic and digital biomarkers and medication response signatures.
PPMI
As highlighted in Objective 1, PPMI utilizes the clinical infrastructure to support biomarker discovery. There are currently over 10 substudies leveraging PPMI infrastructure to pilot novel biomarkers. For example, PPMI substudies are currently supporting the evaluation of a variety of emerging PET imaging tracers as potential biomarker tools, including alpha-synuclein tracers (such as SPAL06 and Modag01), inflammation markers (such as DPA714), and AV133 for vesicular monoamine transporter type 2 (VMAT2) imaging.
Core to ASAP’s mission is ensuring that any research we fund is accessible to the research community at large. Below are examples of lab materials that were successfully deposited as community resources in 2025.
Rodent Models

Alzforum Parkinson’s Disease model database.
ASAP, through the MJFF Tools Program, supported an expansion of the Alzforum Parkinson’s Disease model database to accurately catalog information characterizing preclinical rodent models related to Parkinson’s disease. In 2025, the catalog expanded from 32 to 35 rodent models. The project will conclude with the final entries added in early 2026.
Several mouse models are now available at The Jackson Laboratory (JAX), including a LRRK2 R1441G Conditional Knock-In mouse model (CRN Team Alessi) and DAT-Flp, ANXA1-Cre, A2a-Cre, A2a-FlpO, D1-Cre, and D1-FlpO models (CRN Team Surmeier).
Antibodies
Several Parkinson’s disease targets that ASAP researchers are investigating — including ATP10B, ATP13A2, RILPL2, VPS13C, MHC-I Kb, and MHC-I Kd — lacked commercial antibody tools. Furthermore, antibodies to immunoprecipitate organelles were lacking, including OMP25 (mitochondria), TMEM115 (golgi), and TMEM192 (lysosomes). Antibodies for each of these targets were successfully generated and are undergoing final validation for commercialization in early 2026. Given this success, ASAP funded a new round of antibody development in 2025 to support development of antibodies for iNDI-PD targets, including GRN, CLN3, PSMF1, SMPD1, GALC, DYRK1A, ITPKB, SPTSSB, SLC18B1, and TRPM2.
Cell Lines
iNDI-Parkinson’s Disease (iNDI-PD) is an extension of the original Alzheimer’s disease iNDI project. iNDI-PD focuses on engineering lines that are relevant to Parkinson’s disease or dementia with Lewy bodies. Phase 1 of iNDI-PD focused on developing cell lines with SNVs in well-established disease-relevant genes like SNCA, LRRK2, GBA1, PINK1, and PRKN. Over 15 trio sets have been generated and made available to the research community. This year to date, 147 samples of the cell lines have been distributed to researchers, specifically 114 to non-profit scientists and 33 to scientists in for-profit institutes.

List of iNDI-PD cell lines that are available at JAX as of October 2025. Cell lines that do not have a full trio set are marked with an asterisk (*).
Immortalized cell lines are important resources for high-throughput screening studies and antibody validation. In 2025, ASAP-funded immortalized VPS13C knockout and ATP10B knockout cell lines became available to the research community and supported antibody validation for the targets. An additional two LRRK2 immortalized cell lines passed QC/QA and will be available in early 2026. To support the expanded antibody development efforts mentioned above, ASAP contracted an additional six knockout cell lines in Q3 2025.
Biosamples
PPMI biosamples continue to serve as a valuable resource for the community, supporting biomarker discovery and validation. Over 4,000 participants are enrolled in 50 medical centers across 12 countries, with over 47,000 participants enrolled online. They have completed assessments and shared their data and biological samples, including UPSIT, lumbar puncture, imaging scan (DAT and MRI), skin biopsy, blood, urine, cognitive testing, and more. If you would like to have access to PPMI biosamples, submit a request.

PPMI biosamples request process.
The ASAP Open Science Policy requires grantees to share recipe-style protocols for every method within a manuscript. As of November 2025, over 1,800 ASAP-affiliated protocols were made publicly available on protocols.io, comprising over 5% of the protocols in this database. Engagement with these outputs is high, with some protocols ranked in the top 100 most viewed on the site (over 6,000 views) and others actively receiving multiple bookmarks from users.
In 2025, GP2’s Data Analysis Working Group continued to add to the functionality of our quality control QC pipeline, pushing updates to the public GenoTools GitHub so researchers can benefit from the ongoing improvements, allowing users to apply the same QC metrics and ancestry estimation methods used for GP2’s public release data to their own data. In collaboration with the Data and Code Dissemination Working Group, all researchers make their code available on the GP2 GitHub for the ongoing GP2 core analyses, enabling GP2 researchers to replicate analyses like the new GP2 European ancestry meta-analysis and apply the same analysis pipelines to other data.
In 2025, we continued to promote accessibility of ASAP-funded research outputs by creating 11 videos that deep dive into timely topics. Subscribe to our YouTube channel for the latest videos.

Our Discover ASAP video series highlighted interviews with ASAP-funded researchers discussing their novel outputs, including datasets, code, lab materials, and protocols.
We launched a new Tools and Resources video series to share helpful tutorials for using tools and resources developed by ASAP-funded researchers.
We are committed to openly sharing all ASAP contributions with the research community with new outputs monthly. Thanks to our community’s hard work, the ASAP Catalog has reached a major milestone — over 500 articles shared publicly!
As of November 2025, the Catalog features over 2,800 outputs and tools from our supported programs and research stakeholders, including more than 530 articles, 160 codes, 380 datasets, 620 lab materials, and 1,200 protocols.

 In 2024, ASAP provided support for the Parkinson’s Foundation to expand their PD GENEration: Mapping the Future of Parkinson’s Disease Study, which provides genetic testing and counseling to people with Parkinson’s disease. To date, PD GENEration has enrolled over 30,000 participants, with over 9,500 participants joining in 2025.
In 2024, ASAP provided support for the Parkinson’s Foundation to expand their PD GENEration: Mapping the Future of Parkinson’s Disease Study, which provides genetic testing and counseling to people with Parkinson’s disease. To date, PD GENEration has enrolled over 30,000 participants, with over 9,500 participants joining in 2025.
The GP2 Return of Results Interest Group has developed training modules for the GP2 learning platform in September. The GBA1 case study module was released this fall, and modules for other Parkinson’s disease-relevant genes will be added by the end of 2026.
In 2024, PPMI launched a return of research results information effort for participants related to their DaT imaging and UPDRS scores. Over 2,000 participants enrolled to receive return of result information. In September 2025, PPMI began piloting additional return of results in the U.S. related to UPSIT (loss of smell) percentile scores. Based on user feedback, PPMI plans to roll this out to all PPMI locations and participants.
Building the future of Parkinson’s research begins with supporting the people behind it. This year, we expanded our efforts to build a strong, diverse, and global research community — one equipped with the training courses, workshops, and dependent care support needed to drive discovery forward. From new funding opportunities to targeted support for those balancing research and caregiving responsibilities, we are committed to creating an environment where promising ideas and talent can thrive.
CRN
CRN trainees were actively involved in the 2025 CRN Collaborative Meeting. Trainees had the opportunity to participate in hands-on open science training workshops focused on implementing the ASAP Open Science Policy and data sharing best practices using real datasets provided by ASAP trainees. Additionally, trainees could participate in a workshop that provided a step-by-step demonstration on accessing, exploring, and a hands-on exercise to analyze data within the CRN Cloud Platform. Trainees were also encouraged to present their work during poster sessions and answer researchers’ questions. Selected trainees gave a short talk to present their findings to the network.

2025 CRN Collaborative Meeting poster prize winners.
The CRN launched Team Training & Development, an ASAP-run resource team for ASAP trainees and emerging professionals, offering opportunities for skill-building and career exploration, while fostering connections within the research community, and promoting fellowships, funding, and job opportunities. The goal of Team Training & Development is to provide support and resources to the next generation of Parkinson’s disease researchers.
ASAP launched the  CRN Discovery Fellowship, a competitive award designed to elevate postdoctoral research fellows on the path to independent research careers, foster collaboration across disciplines, and facilitate trainee development. Awarded fellows will receive support to conduct bold, creative trainee-led projects that leverage inter-team collaborations, promote training and mentorship opportunities for trainees within the Network. Stay tuned for the announcement of awardees in 2026!
CRN Discovery Fellowship, a competitive award designed to elevate postdoctoral research fellows on the path to independent research careers, foster collaboration across disciplines, and facilitate trainee development. Awarded fellows will receive support to conduct bold, creative trainee-led projects that leverage inter-team collaborations, promote training and mentorship opportunities for trainees within the Network. Stay tuned for the announcement of awardees in 2026!
GP2

GP2 Micro e-Learning Program.
This year, GP2 launched the Micro e-Learning Program, offering a series of under 10-minute videos that simplify essential concepts in Parkinson’s disease, covering clinical aspects, genetics, data analysis, and GP2 resources. These concise videos are crafted to maximize accessibility and engagement, providing quick, high-impact learning to help viewers stay informed on foundational concepts and recent advancements in Parkinson’s research.
GP2 utilizes a “Train the Trainer” (TTT) model to build local expertise and skills in Parkinson’s disease genetics through ongoing training of local researchers in the GP2 network. This was effective in training workshops delivered in Bucharest, Romania, and Querétaro, Mexico, which were fully led by two GP2 trainees each, as well as workshops delivered in Santiago, Chile, and Accra, Ghana, with one GP2 core member and one GP2 trainee.

GP2 hosted its first “Train the Trainer” (TTT) workshop in May.
GP2 held its third Hackathon in Lübeck, Germany, in August, designed to bring trainees together for hands-on learning, collaboration, and mentorship. 16 trainees participated in intensive project work, using the latest GP2 data with interactive training and networking activities. Several teams used the Verily Workbench to access GP2 datasets and run their analyses directly in a cloud-based environment. Trainees also had the opportunity to network and receive mentoring from some members of the GP2 leadership team, benefitting from conversations covering career development, collaboration ideas, and ways in which GP2 can continue supporting trainees in the future.

GP2 2025 Hackathon group photo in Lübeck, Germany.

GP2 Underrepresented Populations PhD Program promotion.
GP2 received a record number of applications to the 2025 Underrepresented Populations PhD Program, which aimed to support trainees and their supervisors who identify as belonging to an underrepresented group in Parkinson’s disease research. Five applicants were selected for funding from Argentina, Georgia, India, Kenya, and Lithuania. In addition, GP2 supported nine sabbaticals and visiting scientists across the GP2 network this year.
PPMI
Launched in 2024, the Write Now initiative aimed to encourage early career investigators to analyze PPMI results. Awardees are paired with mentors from the PPMI leadership team and connected to the statistical support team to facilitate researchers to analyze PPMI data. Since its launch, 13 projects have been identified, with five already presenting on their findings at conferences and two projects resulting in publications related to safety of lumbar punctures and identifying individuals at-risk of developing Parkinson’s disease using a population-based recruitment strategy.

This year, we expanded our annual Care & Career program — a stipend program that provides resources to offset rising costs of dependent care — to support CRN and GP2 graduate students, early-career researchers, and PPMI clinical coordinators, trainees, and other key personnel. In 2025, we distributed more than 250 stipends of up to $10,000 per household to support child and elder care.
Each month, we continued to recognize individuals within the ASAP CRN in two ways. The Open Science Champion is awarded to individuals who have demonstrated a strong commitment to open science. The Network Spotlight is awarded to individuals for their contributions to ASAP.

In 2025, seven researchers and three CRN teams were recognized as Open Science Champions, and 10 researchers and three CRN teams were nominated by the network through Network Spotlights. We hope others will be inspired to follow their commitment to open science and collaboration.
The work highlighted in this report reflects a collective effort to build upon the foundations that we have laid out over the past five years and to continue to move the needle forward for Parkinson’s research. We are excited to continue making strides in 2026.
Scientific progress thrives when collaboration takes precedence over competition, enabling researchers to share insights, align efforts, and accelerate discovery together. We are endlessly appreciative of our amazing partners who make our vision possible. We thank our implementation partner, The Michael J. Fox Foundation, and the following collaborating organizations: Allen Institute, Bridge Analytics, CatalystNeuro, DataSeer, DataTecnica, DNAStack, ICOR, Parkinson’s Foundation, Verily, and YLD. We can’t wait to see what’s to come in 2026.


























