
ASAP Q1 2026 At-A-Glance

Welcome to the ASAP Research Round-Up Newsletter, your one-stop shop for ASAP updates! Throughout the first quarter of 2026, Aligning Science Across Parkinson’s programs continued to produce resources for the scientific community, support venues that facilitate collaboration, and provide new opportunities for researchers to accelerate discoveries in Parkinson’s disease (PD) research. The image below provides a high-level overview of what we’ve been up to throughout the first quarter of 2026. Want more information? Read the full newsletter for an in-depth look.

In the first quarter of 2026, ASAP programs were involved in more than 25 articles, many of which contributed to our understanding of PD genetics, neuroinflammation, endolysosomal and mitochondrial dysfunction, synaptic function, and the potential use of biomarkers for biological staging. For an overview of the latest preprints, check out our ASAP bioRxiv channel. Below, we highlight five major themes of select papers that came out this past quarter.
Studies continue to map the genetic landscape of PD across diverse global populations and link specific genetic variants to disease progression. Recent papers from across the ASAP initiative have expanded our understanding of the genetic architecture of PD and how these genetic risk factors manifest clinically.
- Modeling regulatory variation in PD brain tissue identified over 53,000 high-confidence variants that modulate when and where genes are activated in specific brain cells, enabling researchers to prioritize regulatory variants at PD GWAS loci using sequence-to-function models. By establishing a mechanistic link between non-coding genetic risk and cell-type-specific dysfunction, this framework transforms static genetic maps into a functional blueprint for identifying the causal pathways driving disease risk. (CRN Team Voet | View preprint)
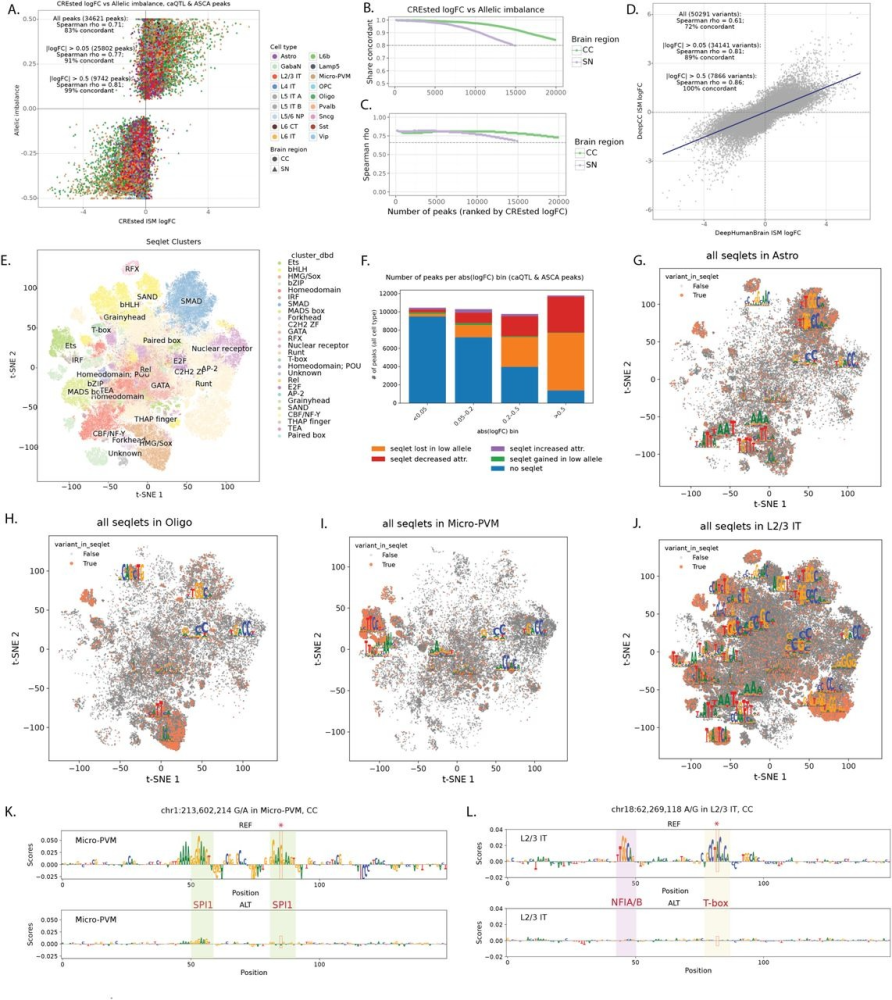
- A comprehensive genetic screening of patients in a South African PD study using the NeuroBooster array identified 16 unique missense variants and novel copy-number variants in genes such as PRKN and SNCA. While the genes involved are globally recognized as major players in PD, the specific variants identified are uncommon in individuals of European ancestry, highlighting the importance of genetic research in underrepresented populations. Mapping these unique mutations is essential for a more complete understanding of the molecular mechanisms driving neurodegeneration and is a vital step toward improving the accuracy of global genetic diagnostics. (GP2 | View preprint)
Is the age of onset for PD written in our DNA, or shaped by where we live? A recent study of the LRRK2 G2019S variant reveals both nature and nurture contribute to age of onset. Examining global data from diverse cohorts participating in GP2, researchers found that ancestry and country of origin can shift PD onset by nearly a decade. This evidence of a strong gene-environment interaction highlights that genetic risk is not a monolith; it is a dynamic variable shaped by background and location. Integrating these factors into diagnostic frameworks will be essential for identifying the right intervention windows for LRRK2-targeted therapies. (GP2 | View publication)
The traditional view of PD as a localized brain disorder is rapidly evolving. Increasing evidence points to the peripheral immune system and the gastrointestinal tract as key players in PD pathogenesis. By exploring how systemic inflammation and gut-pathology converge, these studies reveal that PD is not just a neurological decline, but a complex multisystem disorder involving a breakdown of communication across diverse biological processes.
- For decades, the scientific community has largely treated Alzheimer’s and Parkinson’s as distinct clinical silos. However, the clinical reality is far more complex: a large subset of patients harbor mixed pathologies. In this study, researchers found that when Tau, amyloid-β, and alpha-synuclein co-pathologies are combined in a mouse model, they don’t just co-exist, but act synergistically to put the immune system into overdrive. Crucially, this hyper-inflammation, as demonstrated by expansion of tissue-resident memory T cells and activated microglia, occurs prior to neurodegeneration, opening up a potential window to target for early therapeutic intervention. (CRN Team Kordower | View publication)

- Single-cell spatial multiomics uncovered shared immune-metabolic dysregulation and iron mishandling at the gut barrier in both PD and inflammatory bowel disease (IBD) biosamples, suggesting a common systemic driver for these conditions. This is further evidence that Parkinson’s is a complex condition with systemic collapse across different biological processes. (CRN Team Liddle | View publication)
- Evidence in PINK1 knockout models reveals that gastrointestinal infections are more than a transient event, but can act as a catalyst for neurodegeneration. Without the PINK1 protein to help regulate mitochondrial quality control, an infection can lead to enhanced release of mitochondrial DNA, which activates cGAS/STING inflammatory pathways and, in cellular models, can contribute to neuronal degeneration. (CRN Team Desjardins | View preprint)
- Gut-initiated alpha-synuclein fibrils injected into the duodenum and antrum of mice can drive central dopamine dysfunction that modulates REM sleep behavior disorder (RBD)-like symptoms. This work highlights how peripheral pathology can directly affect neural circuitry underlying both sleep architecture and dopaminergic function. (CRN Team Kaplitt | View publication)
Dysfunctions in cellular organelles, specifically mitochondria and lysosomes, remain a central focus for ASAP Collaborative Research Network (CRN) teams in understanding the mechanisms of PD pathogenesis and progression. Recent papers from ASAP have detailed how specific PD-associated genetic variants unmask metabolic vulnerabilities and disrupt cellular degradation systems.
- We’ve long known that protein aggregates are hallmarks of disease, but precisely how they destroy the cell’s waste management system isn’t as clear. This paper identifies a Cathepsin-driven mechanism where amyloid formation provides the mechanical force necessary to puncture lysosomal membranes, establishing a structural paradigm for how disease-relevant protein aggregates compromise lysosomal integrity. (CRN Team Harper | View preprint)

- LRRK2 G2019S is a well-known genetic risk factor for PD, but genetics don’t tell the whole story. This study highlights a critical second hit mechanism – the impact of metabolic stress. When LRRK2 G2019S knock-in mice were fed a high-fat diet, they exhibited disrupted purine and energy metabolism, demonstrating that pre-existing metabolic syndrome exacerbates mitochondrial dysfunction. (CRN Team Lee | View preprint)
- A cell-type-resolved protein atlas of brain lysosomes has identified SLC45A1 as a uniquely neuron-specific lysosomal protein. This discovery reclassifies SLC45A1-associated disease – previously thought to be a glucose transporter deficiency at the plasma membrane – as a lysosomal storage disorder linked to iron homeostasis and mitochondrial dysfunction. By correcting this long-standing misclassification, the research establishes a new structural paradigm for how cell-type-specific lysosomal proteomes maintain neuronal health and dictate disease vulnerability. (CRN Team Alessi | View publication)
- Structural remodeling of mitochondrial protein biogenesis machinery under proteostatic stress revealed major conformational changes in the mHsp60 protein folding machine. This discovery provides the first near-atomic, in situ visualization of how mitochondria dynamically reprogram their molecular machinery to prioritize protein folding over translation during stress, offering a precise structural roadmap for developing future therapeutics that could bolster mitochondrial resilience in neurodegenerative diseases. (CRN Team Harper and Team Alessi | View publication)
Changes in neural excitability, neurotransmitter release, and synaptic vulnerability are key contributors to motor and non-motor symptoms of PD. Recent papers from ASAP CRN teams have characterized complex adaptations in neuronal signaling, circuitry, and presynaptic function.
- The anterior dorsolateral striatum was identified as a hotspot for dopamine-acetylcholine interactions, which plays an important role in selectively regulating learning and movement control. This finding adds to a growing body of evidence that neuromodulator interactions are spatially organized and provide insights into an anatomical roadmap for developing therapies that could restore the dopamine-acetylcholine imbalance observed in PD and other movement disorders. (CRN Team Cragg | View preprint)
- Impaired synaptic vesicle loading drives dopamine release deficits in human PD patient iPSC-derived neurons via reduced VMAT2 expression. This work highlights a critical bottleneck in the dopaminergic terminal, where failure to sequester dopamine results in cytosolic accumulation that promotes oxidative stress and neurodegeneration. This discovery shifts the therapeutic paradigm, moving beyond simply restoring dopamine levels toward strategies that optimize dopamine handling and sequestration. (CRN Team Cragg | View preprint)
- In vivo proteomics and CRISPR screening identified disruptions in key synaptic proteins, such as STXBP1, that precede neuronal loss and amplify dopaminergic neuron vulnerability to alpha-synuclein toxicity. Rather than focusing solely on clearing late-stage aggregates, this study asks whether we can also prevent neurodegeneration by fortifying synaptic machinery long before irreversible damage occurs. (CRN Team Calakos | View preprint)
The landscape of PD research is undergoing a paradigm shift, moving from clinical observation toward a biological definition of the disease. Central to this evolution is the Neuronal Synuclein Disease (NSD) framework and the alpha-synuclein seed amplification assay. Recent publications from the Parkinson’s Progression Markers Initiative (PPMI) highlight the critical nature of this transition, showing how biological staging significantly improves our ability to predict progression and differentiate individuals with the relevant biology. Here are a few examples of articles that came out this quarter from PPMI investigators.
- Applying the Neuronal Alpha-Synuclein Disease (NSD) biological staging framework to analyze the five-year progression of sporadic PD participants within the PPMI cohort reveals that a participant’s baseline biological stage strongly predicts the rate of future functional decline. The analysis also highlights significant clinical heterogeneity among patients who are traditionally classified together as early-stage PD patients, underscoring the need to use both biological and functional inclusion criteria in clinical trials. (PPMI | View publication)
- Individuals clinically diagnosed with PD who test negative for the alpha-synuclein seeding aggregation assay in CSF (CSF SAA-negative) frequently exhibit baseline motor and cognitive symptoms that are nearly indistinguishable from those who are CSF SAA-positive. However, the longitudinal data tells a different story – 1 in 7 SAA-negative patients within the PPMI cohort eventually received a revised diagnosis, compared to less than 1% of the SAA-positive group. This biological gap in our current diagnostic criteria suggests that clinical observation alone can mask a variety of underlying pathologies. It also underscores that CSF SAA stratification should be consideredno longer be optional for PD clinical trials, especially those using synuclein-targeting therapies. (PPMI | View publication)
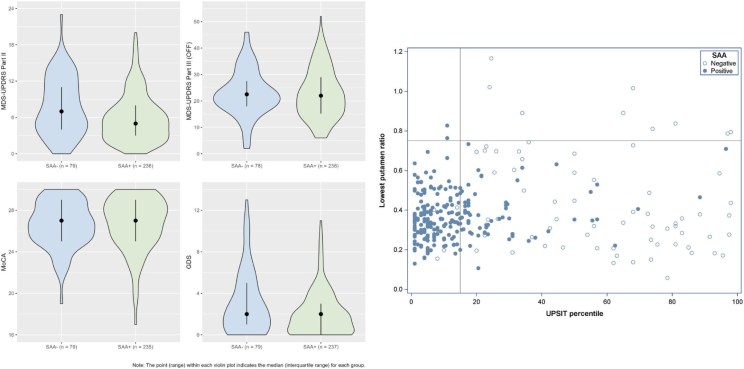
- Does LRRK2-Parkinson truly progress more slowly than sporadic forms of the disease? A recent preprint utilizing PPMI data suggests that for the ~70% of LRRK2 carriers who are CSF SAA positive (S+), the “slower” progression label may be a misconception. While S+ LRRK2-PD patients often present with milder motor symptoms and less dopaminergic deficit at baseline, their four-year clinical trajectory is virtually identical to that of matched sporadic PD patients. It also underscores that CSF SAA stratification should be considered for PD clinical trials, especially those using synuclein-targeting therapies. (PPMI | View preprint)
To continue to improve our understanding of PD, submissions opened for several new RFAs, including:
- Immune Profiling Insights Program | As we move beyond a one-size-fits-all approach to PD, understanding the role of the immune system is critical. Building upon the CRN 2020 neuroinflammation theme and following the 2025 establishment of an Immune Consortium to identify robust immune signatures in PD, The Michael J. Fox Foundation for Parkinson’s Research (MJFF), in partnership with ASAP, is pleased to announce a new funding opportunity through MJFF: the Immune Profiling Insights Program. The purpose of this program is to support research that deepens our understanding of the immune system’s role in PD.
- Identify specific immune cell populations and functional states that shift during the PD continuum.
- Determine how immune features correlate with clinical outcomes and disease progression.
- Inform the development of future therapeutic targets and patient selection strategies.
- Multi-Omics Meta-Analysis | In June 2024, ASAP launched the CRN Cloud, a data-sharing tool that houses omics data from CRN Teams, making this data accessible to the broader scientific community. The data analysis pipeline shouldn’t stop at storage. Instead, we want to encourage CRN teams to look across the data housed within the CRN Cloud to assess replicability, presence of common patterns, and potential for novel mechanistic insights. In 2025, we funded 10 projects to support meta-analyses across CRN Cloud datasets. In Q1 2026, we announced an additional opportunity for CRN Teams to apply for small meta-analysis projects that use data stored in the CRN Cloud with a goal of driving innovation. We look forward to announcing these new projects later this year!
- The PPMI Write Now Initiative | The PPMI Write Now Initiative encourages junior investigators, such as fellows or assistant professors within their first five years of appointment, to engage with PPMI data and publish papers that advance the field. Applications were open through March 30th, 2022026. We look forward to seeing the work that comes from these awardees!

In the first quarter of 2026, ASAP supported virtual and in-person events to facilitate connections and provide avenues for sharing ideas.
CRN Team Hurley hosted three Mito911 Webinars to discuss the latest research in mitochondrial biology, autophagy, and neurodegeneration. During this past quarter, we heard talks from David Chan, MD, PhD (Caltech), Timothy Ryan, PhD (Weill Cornell Medicine), and Elena Ziviani, PhD (Universita di Padova).
- Interested in viewing some of the past Mito911 events? ASAP created a YouTube playlist featuring recordings of past Mito911 webinars to promote connections across the research community. Check it out!
CRN Team Alessi hosts the LRRK2 Central Webinar, which focuses on LRRK2 biology in PD research. This past quarter, we heard from Ji Sun, PhD (St. Jude Children’s Hospital).
PPMI investigators gave more than 10 presentations at AD/PD across multiple sessions, including in next-generation biomarkers across Alzheimer’s, PD, and Lewy Body Dementia (LBD); mechanistic insights into PD and LBD pathology; and predicting PD and LBD: biomarkers and beyond.
126 investigators, clinicians, and staff attended the GP2 Regional Meeting from March 9-10 in Nairobi, Kenya. Over 25 countries were represented, including 15 countries within Africa. In addition to updates on ongoing African cohort data collection and analysis, emerging themes from the meeting focused on addressing gaps in African genetic representation and building research capacity in Africa.

ASAP was one of several sponsors for the 2026 GRC Basal Ganglia Meeting, which brought together leading international experts from the US, Europe, Australia, and Asia to share cutting-edge research on neural circuitry and brain function. In addition, ASAP sponsored the 2026 GRC Autophagy in Stress, Development, and Disease, which provided a platform for researchers studying autophagy mechanisms to connect with experts in aging, neurodegeneration, and other disciplines.


Promoting open science is at the core of all ASAP work. We are committed to sharing our philosophy and strategy for implementing our Open Science Policy. Some highlights of our engagement with the broader open science community from the first quarter of 2026 include:
Robert Thibault, PhD, from the ASAP Open Science Team, sat down with the Science Integrity Alliance to discuss ASAP’s Open Science Policy and the support we offer our grantees to successfully implement it. Read more starting on page 48.

Maria Machado & Luciana Machado. A Funder-Led Approach to Open Science. REACH; January–March 2026: 48–53. Available at: https://www.sci-integrity.com/reach-january-march-2026
CRN Cloud Data Releases: In Q1 2026, CRN Cloud added 10 new datasets and expanded the supported sample sources to include gastrointestinal tissue, liver tissue, fecal samples, and embryonic fibroblasts. This quarter marked the first time metagenomics data was released on the CRN Cloud. Q1 2026 releases also included additional proteomics, bulk RNA-seq, single-nucleus RNA-seq, and spatial transcriptomics datasets. These new datasets added 5,092 new samples across 684 new subjects, including 530 human donors (liver, fecal, and colon).
Interoperability of CRN Cloud with AMP® PDRD and GP2: The CRN Cloud launched a new integration with Accelerating Medicines Partnership Parkinson’s Disease and Related Disorders (AMP® PDRD) and GP2 to support cross-program analyses and new discovery opportunities. The integration enables users to analyze CRN Cloud datasets alongside AMP® PDRD and GP2 within a single Verily Workbench workspace, which makes it easier to combine CRN omics data with large-scale genetics and cohort datasets.
To access these datasets in a shared workspace, researchers must have approved access to each platform individually and use the same email account across platforms. Detailed setup instructions are available in a step-by-step user guide, linked directly from the CRN Cloud collection cards below:
Natural History Study: PPMI continues to support data access to a full breadth of individual-level PPMI data, including clinical, imaging, ‘omics, genetic, sensor and biomarker data. As of this last quarter, there is data on 2,864 PD participants, 4,317 prodromal participants, and 574 healthy controls. Learn more.
Antibodies: The first ASAP-funded antibodies were made available!
- ATP10B
- Human Reactive (WB)
- ATP13A2
- Human Reactive (WB)
- Human Reactive (ICC/IF, IHC-P, IP, WB)
- Mouse Reactive (ICC/IF, IHC-P, WB)
- OMP25
- Human, Mouse Reactive (IP, WB)
- RILPL2
- Human, Mouse Reactive (ICC/IF, IP, WB)
- Mouse Reactive (ICC/IF, IP, WB)
- TMEM115
- Human Reactive (IP, WB)
- TMEM192
- Human Reactive (unconjugated) (Elisa, IP, WB)
- Human reactive (bead conjugated) (IP)
- VPS13C
Cell Lines: Human-derived cell models are a critical research tool that enables biology and preclinical therapeutic studies in a human genetic context. The following lines were released this quarter, allowing researchers to interrogate the role of targets in cell health and PD.
- Hear more about Automated Quantification of Intracellular Neuromelanin in Brain Tissue from Anastasia Filimontseva (CRN Team Vila)
- Discover Dr. Armin Bayati’s (CRN Team Chen) AutoMorphoTrack: A Platform for Quantitative Organelle Analysis
- Learn more about A Programmable Self-Assembling System to Model Intracellular Protein Aggregation with Dr. Yujie Fan (CRN Team Gradinaru)

In the first quarter of 2026, ASAP enabled researchers to expand their knowledge through a variety of training opportunities.
In January, GP2 marked a major milestone with the opening of Nigeria’s first dedicated molecular facility at the College of Medicine, University of Lagos College (CMUL). Led by Professor Njideka Okubadejo, this GP2-supported laboratory will serve as both a DNA extraction and biobanking facility, enabling local processing of samples for submission to GP2 and serving as a training location for students and researchers.
As CMUL leadership noted, “Talent is not the constraint in Nigeria; it is the platform and opportunity. The new laboratory creates both.”
In addition to its biological applications, the facility will serve as a training hub, providing students with stipends to support their participation and reduce financial barriers to research experience. GP2 is proud to play a part in strengthening local research infrastructure, supporting formal training and capacity building for researchers.

In March, GP2 released a new micro e-learning video, “Identifying and Phenotyping Individuals At-Risk for Parkinson’s Disease | Micro e-Learning-Clinical.” The video covers recruitment strategies, remote assessment tools, and how digital health technologies are expanding research beyond traditional clinical environments. It also highlights key considerations such as participant engagement, communicating genetic risk, and navigating new diagnoses.
We are pleased to announce the launch of CRN Team Training & Development! This Team provides CRN trainees and emerging professionals opportunities for skill-building and career exploration, fosters connections within the research community, and promotes fellowships, funding, and job opportunities. The Team officially launched in March, with a career workshop featuring Shelby Johnson, PhD, from the Coalition for Aligning Science. During this workshop, Dr. Johnson discussed her career path, including her time as a senior scientist at a pharmaceutical company.

Did you know that the GP2 website has a list of career opportunities? Check them out here!

